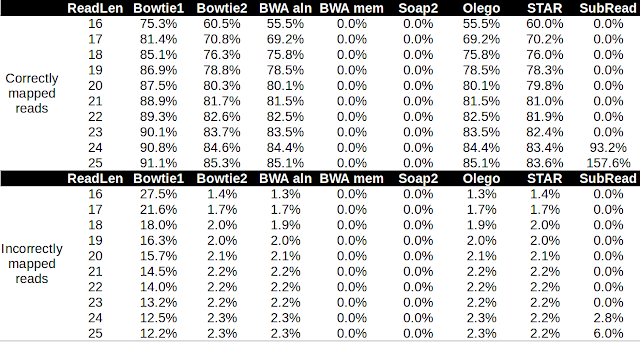
2014 Wrap-Up

The year has gone so fast! Lets go through some of the major points of 2014 and predict what 2015 has in store. Sequencing hardware It began with announcements from Illumina of the HiSeqX10 as well as the release of the NextSeq500. Later, X-10 technology was included in V4 HiSeq2500 kits resulting in a substantial increase in sequence output from that instrument. There was also the announcement of the NeoPrep automated library preparation system that will be officially released in the 1st half of 2015. NeoPrep looks like an attempt to use microfluidics to perform generate libraries; if this is successful, it would be a major advance in reducing bottlenecks in sample preparation. Microfluidics are also able to reduce the volumes of reagents required, making the process cheaper. In addition, labour costs per library will be drastically reduced given that automation will be commonplace for routine protocols. Regarding 3rd gen platforms, we saw some mixed reviews from Oxford Nanopore ...